Pompano Broodstock Maturation and Larviculture Protocols

Slide-1
Pompano Broodstock Maturation and Larviculture Protocols
Slide-2
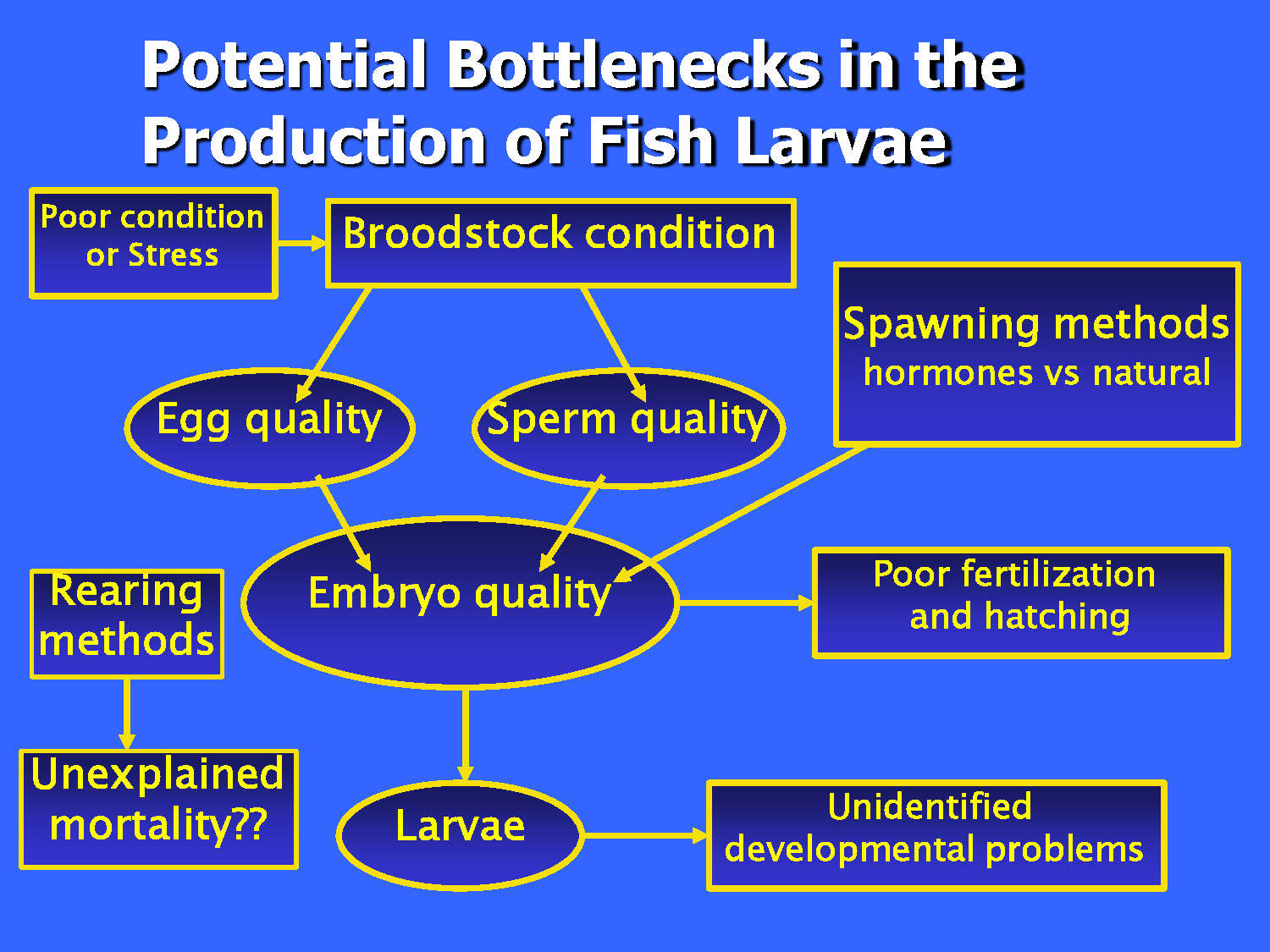
A flow chart titled "Potential Bottlenecks in the Production of Fish Larvae"
The flow begins at the top left with a box labeled "Poor condition or Stress," which points to "Broodstock condition."
From "Broodstock condition," two arrows lead down to two ovals: "Egg quality" and "Sperm quality."
Both quality ovals, along with a box titled "Spawning methods (hormones vs natural)," point toward a central large oval labeled "Embryo quality."
Three paths lead away from "Embryo quality":
- An arrow points right to a box labeled "Poor fertilization and hatching."
- An arrow points down to an oval labeled "Larvae."
- From the "Larvae" oval, an arrow points right to a box labeled "Unidentified developmental problems."
On the far left, a separate vertical flow shows a box labeled "Rearing methods" pointing down to a box labeled "Unexplained mortality??"
Slide-3
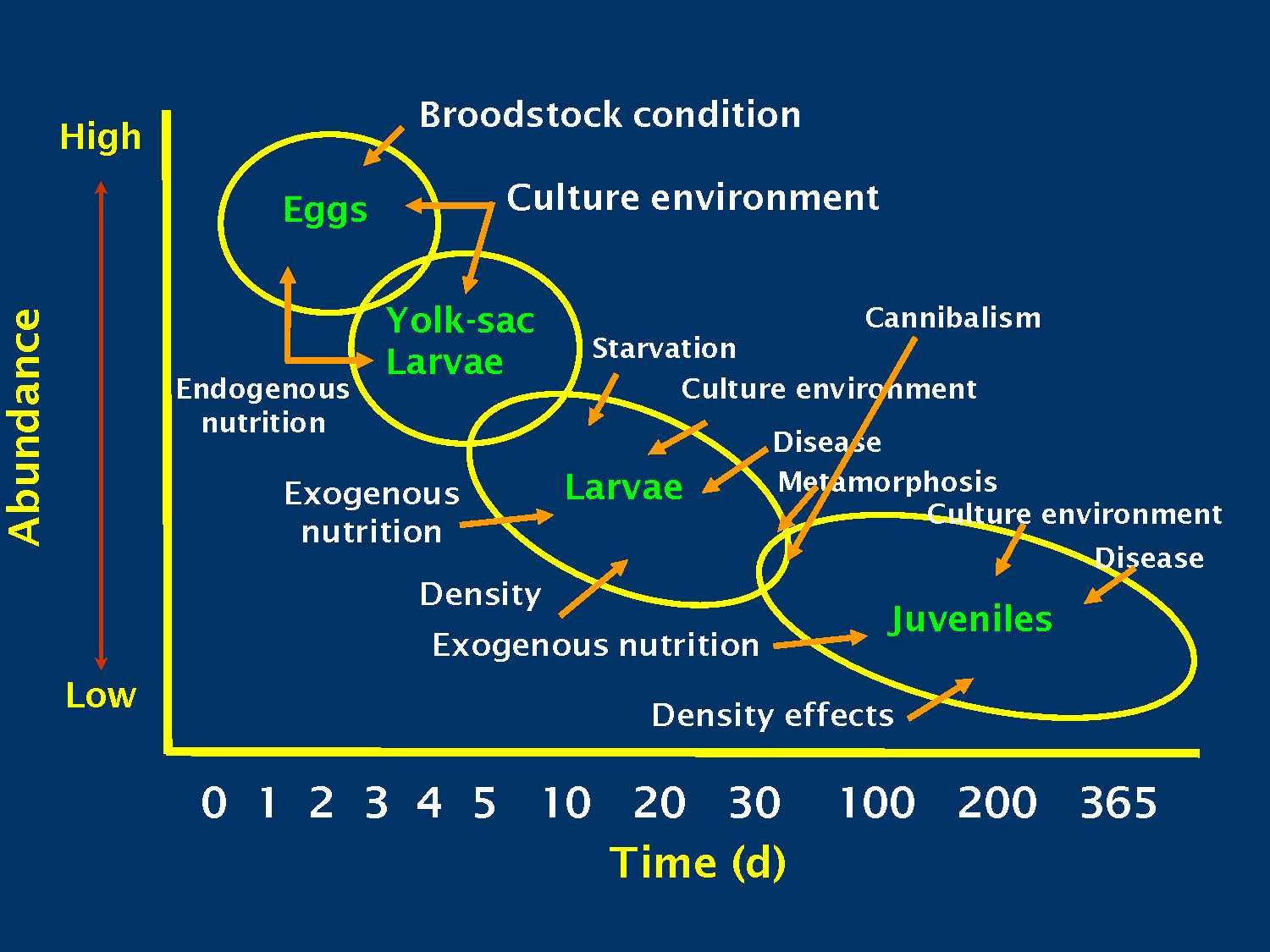
The image shows a graph with a y-axis representing Abundance from Low to High and an x-axis representing Time (d) with increments of 0, 1, 2, 3, 4, 5, 10, 20, 30, 100, 200, and 365.
-
Eggs (located at High Abundance, 0-3 days)
- Influenced by: Broodstock condition
- Influenced by: Culture environment
-
Yolk-sac Larvae (located slightly lower Abundance, 2-5 days)
- Influenced by: Culture environment
- Influenced by: Endogenous nutrition
-
Larvae (located at moderate Abundance, 5-30 days)
- Influenced by: Starvation
- Influenced by: Culture environment
- Influenced by: Disease
- Influenced by: Metamorphosis
- Influenced by: Exogenous nutrition
- Influenced by: Density
-
Juveniles (located at Low Abundance, 30-365 days)
- Influenced by: Cannibalism
- Influenced by: Culture environment
- Influenced by: Disease
- Influenced by: Exogenous nutrition
- Influenced by: Density effects

Slide-4
Broodstock
- Quarantine
- Maturation System Design/Stocking
- Feeding
- Conditioning
- Spawning/Hormone manipulation

Slide-5
Quarantine
- All new fish brought into the facility are isolated until cleared of any and all parasites
- This can take up to several weeks
- Disease/acclimation procedures
- Weening/acclimation procedures

Slide-6
Disease/Acclimation Procedures
- Pompano will always come in with capture wounds
- Immediately treat with a freshwater dip for 5 minutes
- After one week, a prophylactic treatment of formalin (250ppm for 1 hour)
- If disease event occurs after this, determine disease agent and treat accordingly (e.g., Amylo- .20-.25ppm Cu for 3 weeks)

Slide-7
Weening/Acclimation Procedures
- Pompano will typically not eat for the first week in captivity
- To get them started, use frozen krill
- Ultimately, you want to get them on the maturation diet food mix

Slide-8
System Design
Tanks should be supplied with own air/LOX and water inlets so that individual tanks can be isolated if chemical treatments are necessary
Recirculating System Components:
- Biofilter
- Cartridge filtration
- UV sterilization
- Temperature control (heat pump)
- Lighting control

Slide-9
Broodroom System
Image shows multiple circular broodstock tanks arranged in a facility with controlled lighting and water systems.

Slide-10
Broodroom System
Image displays overhead view of broodroom showing tank arrangement, piping, and support equipment for the recirculating system.

Slide-11
Stocking
- Ratio of males to females in the brood tanks is typically 1:1 (2:1 is also an option)
- Female pompano target size 3-6 pound range
- Male pompano target size 1-3 pound range
- Our maturation tanks are approximately 1000 gallons stocked with 24 fish (12 males, 12 females)

Slide-12
Feeding
- Pompano have a high energy requirement
- Fish are fed 3-5% of the total biomass in the tank per day
- Feeding frequency is twice a day, morning and late afternoon feedings
- Maturation diet is an enriched powder mix (e.g., Florida Aqua Farms- Gelly Belly Food Mix)

Slide-13
Conditioning
- This allows the operator to manage broodstock maturation through photo/thermal manipulation
- Temp/light cycles are changed every week
- It is important that the cycle not be interfered with once started
- If a disease event occurs, hold the fish at the current stage and do not advance any further
- Once they are healthy and eating well, cycle will commence
Slide-14
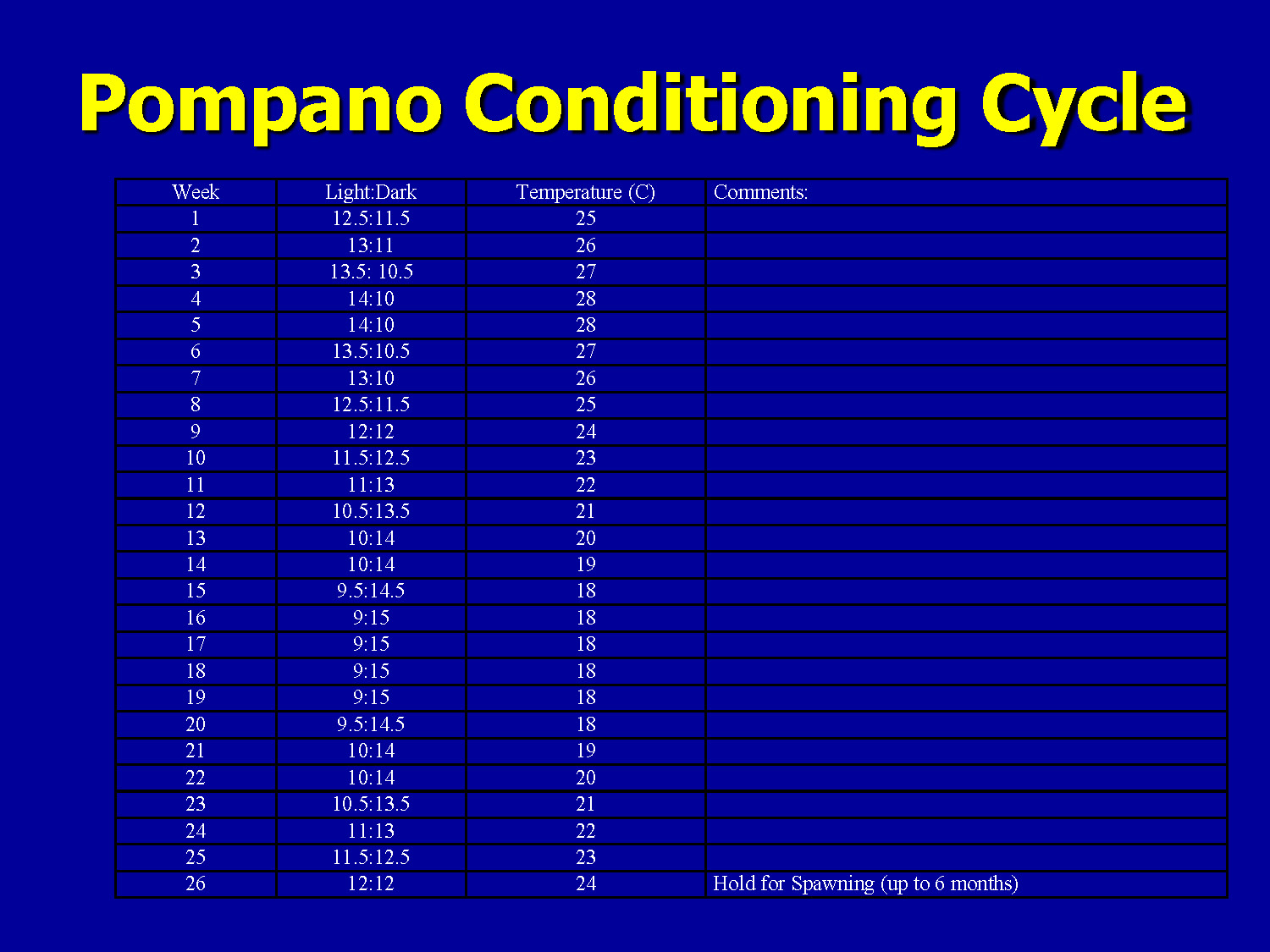
Pompano Conditioning Cycle
| Week | Light:Dark | Temperature (C) | Comments: |
|---|---|---|---|
| 1 | 12.5:11.5 | 25 | |
| 2 | 13:11 | 26 | |
| 3 | 13.5: 10.5 | 27 | |
| 4 | 14:10 | 28 | |
| 5 | 14:10 | 28 | |
| 6 | 13.5:10.5 | 27 | |
| 7 | 13:10 | 26 | |
| 8 | 12.5:11.5 | 25 | |
| 9 | 12:12 | 24 | |
| 10 | 11.5:12.5 | 23 | |
| 11 | 11:13 | 22 | |
| 12 | 10.5:13.5 | 21 | |
| 13 | 10:14 | 20 | |
| 14 | 10:14 | 19 | |
| 15 | 9.5:14.5 | 18 | |
| 16 | 9:15 | 18 | |
| 17 | 9:15 | 18 | |
| 18 | 9:15 | 18 | |
| 19 | 9:15 | 18 | |
| 20 | 9.5:14.5 | 18 | |
| 21 | 10:14 | 19 | |
| 22 | 10:14 | 20 | |
| 23 | 10.5:13.5 | 21 | |
| 24 | 11:13 | 22 | |
| 25 | 11.5:12.5 | 23 | |
| 26 | 12:12 | 24 | Hold for Spawning (up to 6 months) |

Slide-15
Spawning/Hormone Manipulation
- Pompano are spring spawners with an ideal spawning temp of 24°C and 12 hr light
- Natural spawning of pompano in captivity is unreliable
- Hormone manipulation is necessary for consistent and controlled spawning
Slide-16
Spawning/Hormone Manipulation
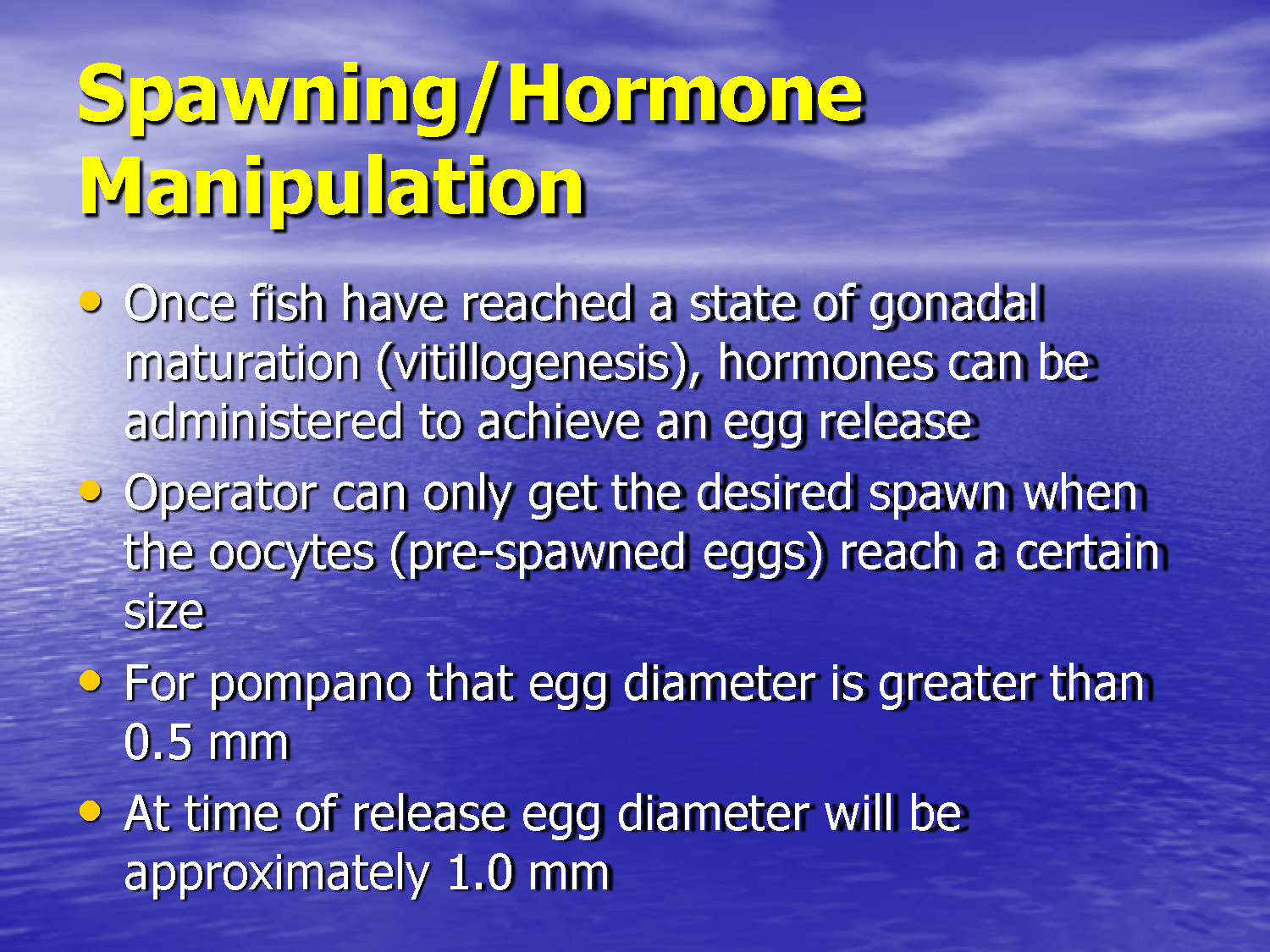
- Once fish have reached a state of gonadal maturation (vitillogenesis), hormones can be administered to achieve an egg release
- Operator can only get the desired spawn when the oocytes (pre-spawned eggs) reach a certain size
- For pompano that egg diameter is greater than 0.5 mm
- At time of release egg diameter will be approximately 1.0 mm

Slide-17
Spawning/Hormone Manipulation
- Salmon Gonadotropin releasing hormone analogue (sGnRHa) – 75 µg dose
- This is a slow release implant which gives the fish an initial burst of gonadotropin followed by several days of lower levels
- Pompano is a high energy fish, however, and will usually yield one viable spawn two days (36hrs) after injection of implant
- Any other residual spawns are typically not viable

Slide-18
This image consists of three photographs detailing the process of harvesting fish eggs (likely pompano) in a hatchery setting.
Top Left Photo: A blue rectangular tank equipped with white PVC plumbing. Water flows from a large pipe through a blue valve into a fine-mesh collector bag suspended inside the tank. The bag is held in place by orange clamps and white support rods.
Bottom Left Photo: A close-up view of the water's surface inside the collector. Small, translucent, spherical fish eggs are visible floating amongst bubbles and foam, indicating a successful spawn collection.
Right Photo: A person’s arm and hand are shown lifting the rectangular mesh collector bag by its support rods. The bottom of the bag tapers into a small PVC fitting, which is being positioned over a blue plastic bucket to drain and concentrate the harvested eggs.
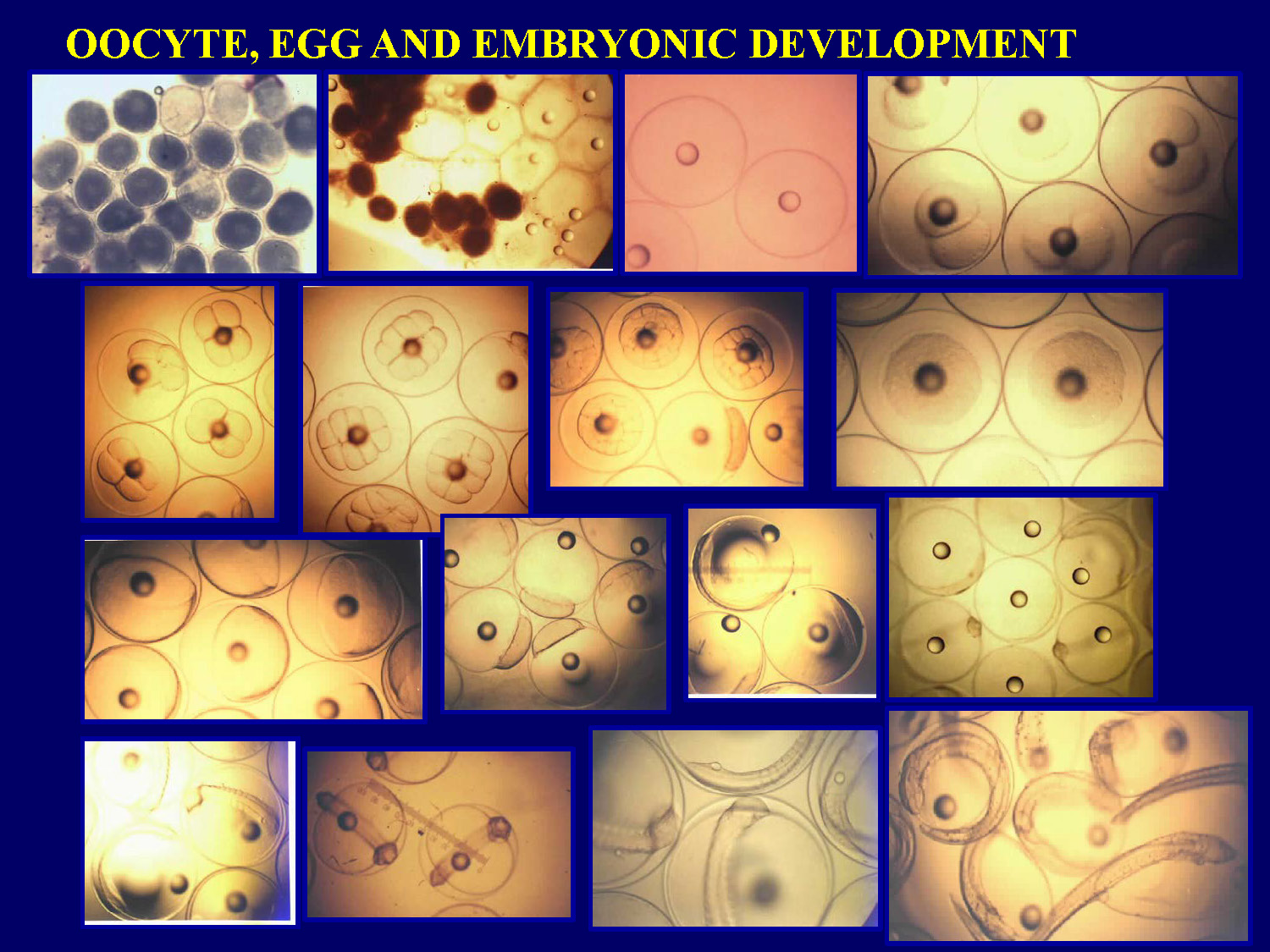
Slide-19
OOCYTE, EGG AND EMBRYONIC DEVELOPMENT
Diagram illustrates the developmental stages from oocyte through fertilized egg to embryonic development in pompano.

Slide-20
Larviculture-Hatching
- Unfertilized eggs can contribute to the fouling of a hatching container
- This can be reduced by ensuring proper fertilization
- Removal of unfertilized eggs will reduce potential for fouling

Slide-21
This image consists of three photographs showing the process of separating and measuring fish eggs within a hatchery facility.
- Top Photo: A technician wearing an orange cap and blue shirt is shown in a hatchery aisle. He is pouring water and collected eggs from a white plastic bucket into a clear, cone-shaped separator (Imhoff cone) supported by a white PVC frame.
- Bottom-Left Photo: The technician is crouched beside the conical separator. He is turning a red valve at the narrow bottom of the cone to drain out cloudy water and non-viable eggs (which have settled at the bottom) into a small plastic container, leaving the healthy, floating eggs behind.
- Bottom-Right Photo: A close-up view of a clear graduated cylinder. The cylinder contains water with a dense layer of small, translucent fish eggs concentrated at the top, allowing for an accurate volumetric measurement of the total egg count.

Slide-22
Larviculture-Hatching
Disinfectants for keeping bacteria and fungus off eggs:
Betadine, methylene blue, formalin, nitrofurazone, malachite green, perioxide
With proper water quality, chemicals are rarely needed

Slide-23
Larviculture-Counting of Eggs
- An egg count should be approximated prior to stocking to determine hatch rate and to ensure proper stocking density
- Obviously you are not going to hand count each egg
- Rule of thumb 1 mL of pompano eggs is approximately 1000 eggs (e.g., 40 mL is 40,000 eggs)

Slide-24
Four images detailing egg development and incubation.
- Top-Left: Microscopic view of four fish eggs showing early-stage embryos curled around the yolk.
- Top-Right and Bottom-Left: Interior views of a hatchery room featuring large black circular incubation tanks with aeration tubes and bubbling water.
- Bottom-Right: Microscopic view of newly hatched, elongated larvae.

Slide-25
A close-up photograph of a large, clear graduated plastic pitcher. The water inside is densely packed with thousands of tiny, pepper-like specks, which are newly hatched pompano larvae being prepared for transfer to larviculture tanks.

Slide-26
Intensive Tank Culture Larval Rearing Systems
- High stocking densities (50-150 larvae/liter)
- High water exchange rate (minimum 100% turnover rate daily) or intensive recirculating systems
- Live microalgae/microalgae paste (Nannochloropsis spp; "green water")
- Rotifers (Brachionus sp. enriched)
- Artemia (Nauplii + Metanauplii enriched)
- Artificial weaning diets (dry feeds)

Slide-27
Larval Rearing Tanks
- Dark-walled tanks preferred because larvae can see prey better against a dark background.
- Tanks should allow easy viewing, feeding, treatment, and harvesting
- Tank volume: 800 Liters
- Larval exclusion screens of different mesh sizes is required to retain larvae during water exchange (e.g., 150, 350 and 500 micron screens)

Slide-28
Three photos showing the scale of a larviculture facility.
- Top-Left: A single large, light-colored circular tank filled with water and equipped with overhead aeration lines.
- Bottom-Left and Right: Wide-angle views of the facility room containing dozens of black circular tanks arranged in rows under fluorescent lighting, interconnected by white PVC plumbing.

Slide-29
A top-down view of a "Larviculture Tank." The tank is black-walled with a light-colored bottom. In the center is a white cylindrical standpipe screen. Two white aeration stones are positioned on the tank floor to provide oxygen and water circulation.

Slide-30
Larval Rearing Systems
Tanks should be supplied with air/LOX and water inlets so that individual tanks can be isolated if chemical treatments are necessary
Recirculating System Components:
- Biofilter
- Particulate filtration to 5 microns or less
- UV sterilization
- Temperature control (heat pump)

Slide-31
An interior view of the hatchery's life support and filtration infrastructure. The image shows:
- Several large, dark green spherical pressurized filter tanks connected by white PVC piping.
- Multiple tall, grey vertical filtration canisters standing on a wooden platform.
- A complex network of PVC pipes, valves, and gauges designed to maintain water quality for the adjacent larviculture tanks visible on the left.

Slide-32
A detailed view of the water treatment and recirculation setup. The image features:
- A large, rectangular white plastic water reservoir serving as a central hub for the filtration system.
- Vertical white UV sterilization units and grey filter canisters integrated into the plumbing line.
- A white tarp or liner suspended overhead to protect the equipment and control light levels within the facility.

Slide-33
Yolk-sac larvae
- Eggs are stocked into larval tanks
- Eggs hatch the following day (day 0)
- Larvae live on yolk sac for 2-3 days after hatching
Image shows microscopic view of yolk-sac larvae with visible yolk sac attached to developing larvae.
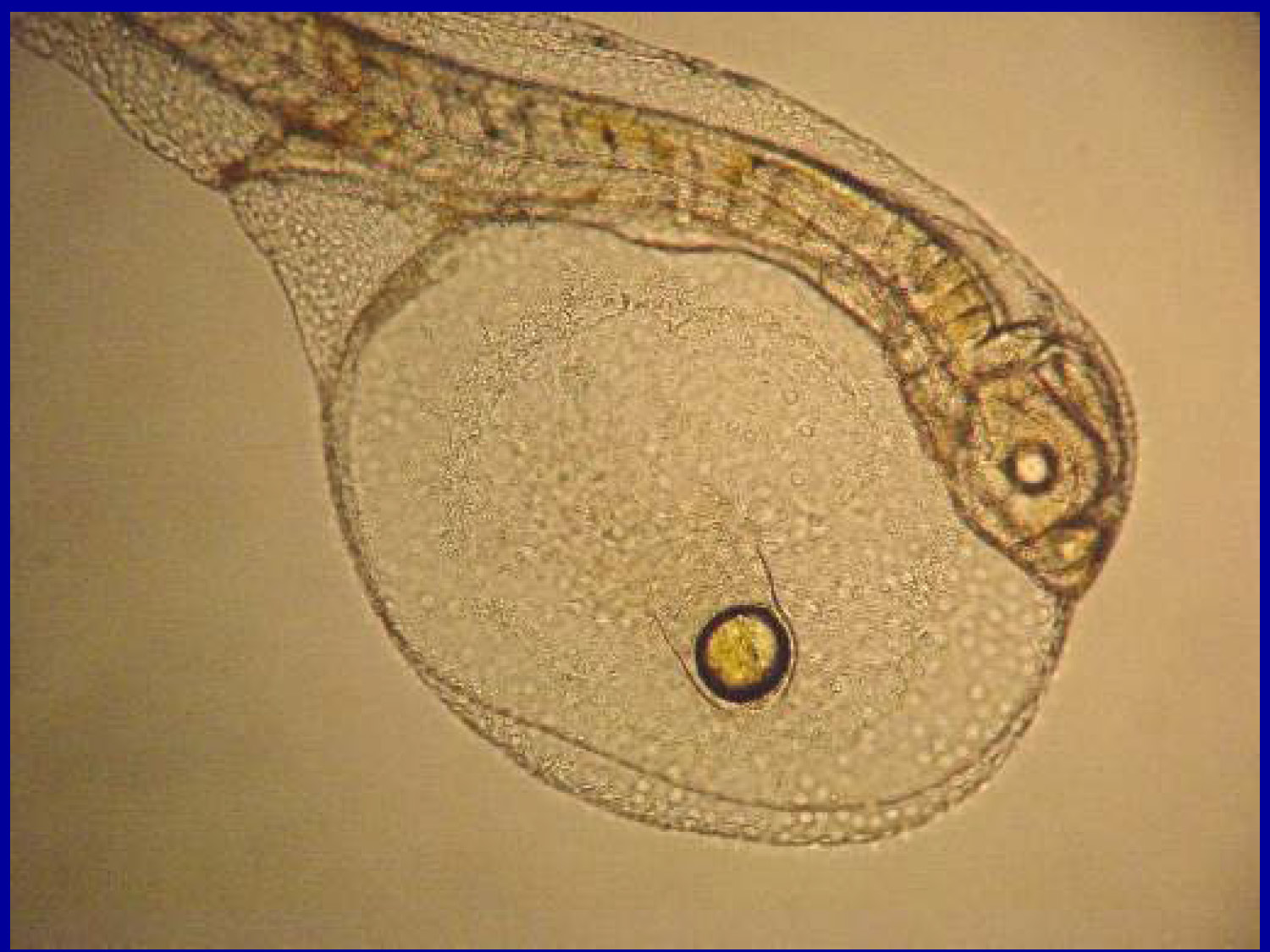
Slide-34
An extreme microscopic close-up of a developing fish larva. It features a prominent, large translucent yolk sac containing a single, dark circular oil globule, which provides nutrition to the larva before it begins active feeding.

Slide-35
LIVE FEEDS (Microalgae, Rotifers, Artemia, Copepods, Plankton) Protocols(maintenance/production/enrichment/disinfection)
Images show three types of live feeds:
- Rotifer: Brachionus spp. - Small zooplankton used as first feed for larvae
- Brine shrimp: Artemia salina - Larger feed for growing larvae
- Phytoplankton: C-Isochrysis sp, Nannochloropsis sp, Diatoms - Microalgae for green water and enrichment

Slide-36
Larval Feeding Protocol for Florida Pompano
- Day 0-2: Green water introduced
- Day 2-15: Enriched rotifers (main feed from day 2-9)
- Day 9-21: Artemia nauplii introduced and continued
- Day 12-21: Enriched Artemia added
- Day 15-24: Microfeeds (dry feeds) introduced and gradually become primary diet

Slide-37
Feed Protocol
- Day 0- Eggs hatch
- Day 2- Green water and 1st feeding-enriched rotifers (3 rotifers/mL up to 6 rotifers/mL)
- Day 9- Start introducing newly-hatched artemia (.25-1 artemia/mL up to 4-5 artemia/mL)
- Day 12- Start introducing enriched artemia (2 artemia up to 4-5 artemia /mL)
- Day 14- Start introducing dry feed along with newly hatched and enriched artemia
- Day 22- Larvae should be weened onto dry feed

Slide-38
Critical Periods During Larval Development
- First Feeding
- Swim Bladder Inflation
- Feed transition
- Metamorphosis
- Weaning

Slide-39
First Feeding
- First feeding is a critical event for larvae
- Upon opening their mouth, the gut is colonized by bacteria. If these are harmful, the larvae will die.
- Fish larvae are visual predators; therefore must offer live zooplankton as first food

Slide-40
Size of First Prey
Size of the initial prey item is dependent upon mouth gape
- Cod - Ciliates, trochophores
- Grouper, snapper – small copepods
- Pompano, cobia – rotifers
- Hybrid striped bass – Artemia
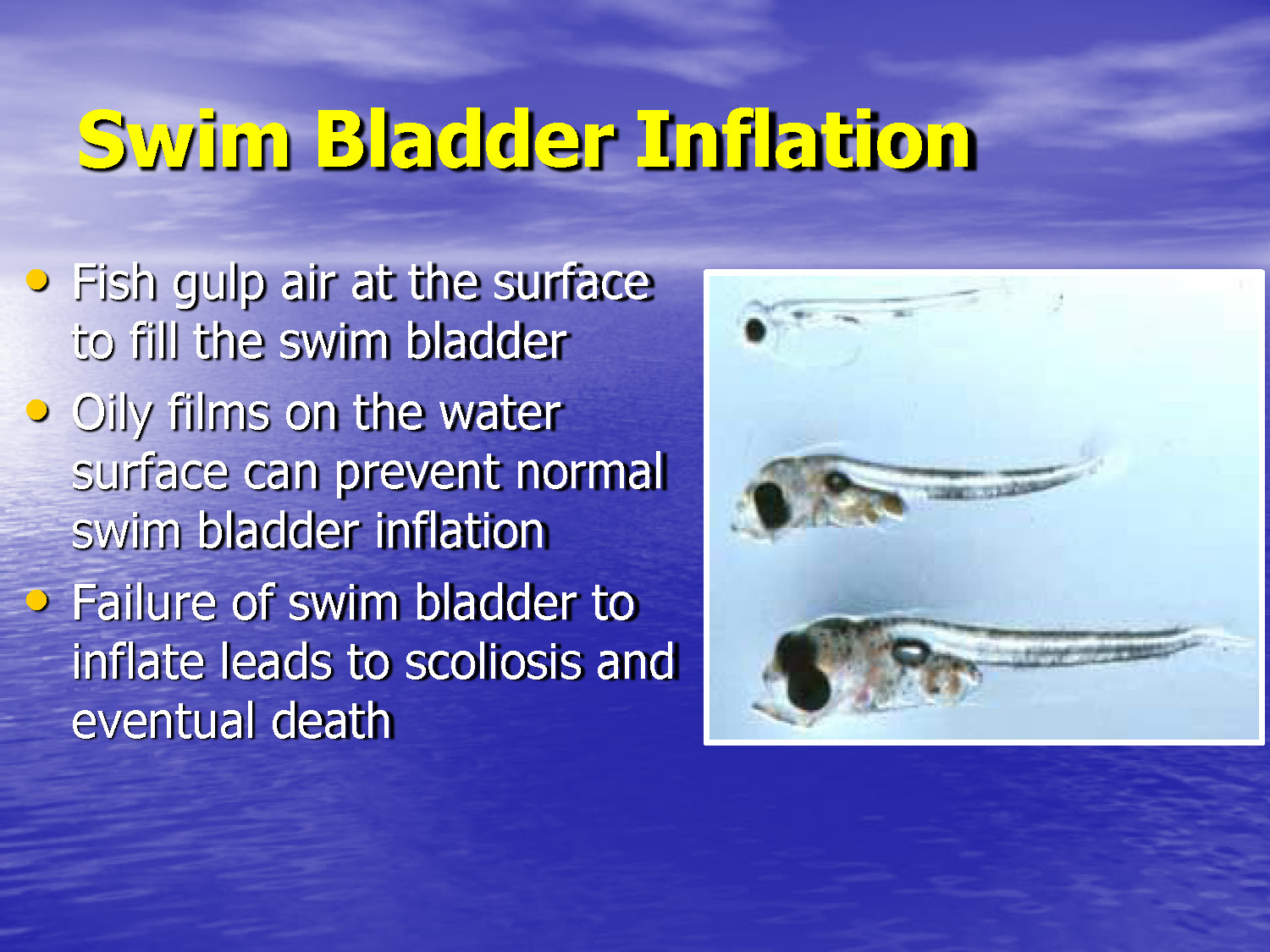
Slide-41
Swim Bladder Inflation
- Fish gulp air at the surface to fill the swim bladder
- Oily films on the water surface can prevent normal swim bladder inflation
- Failure of swim bladder to inflate leads to scoliosis and eventual death

Slide-42
Feed Transition
A photograph titled "Feed Transition." It shows a microscopic view of a pompano larva's head with its mouth open, positioned to strike at a nearby rotifer and a brine shrimp nauplius floating in the water.

Slide-43
Metamorphosis
Metamorphosis is the physical transformation of the fish from its larval form to its juvenile form
Metamorphosis can be negatively impacted by:
- Dietary deficiencies
- Inappropriate temperature regimes (29°C)
- Poor water quality (D.O.-100%, pH-7.0-7.7, Salinity-35ppt, Alkalinity >100mg/L, TAN<.5mg/L, Nitrite<1.0mg/L)

Slide-44
A close-up of a developing juvenile pompano. The fish is becoming more opaque and silver, showing a defined eye and fins. It is surrounded by small orange-colored brine shrimp (Artemia) in the water column.

Slide-45
Weaning
- Weaning is often difficult
- Artificial feeds need to be introduced early, even before fry begin feeding on them so that they are "part of the environment"
- Weaning failure often leads to cannibalism

Slide-46
Bottom line
Two images under the heading "Bottom line."
- Left: A green mesh net containing dozens of small, silver juvenile pompano ready for stocking.
- Right: Several large, adult-sized pompano displayed on a bed of crushed ice, representing the final marketable product.

Slide-47
Vendor List
| Vendor | Item |
|---|---|
| INVE | Artemia Cysts, Sparkle (rotifer supplement), A1 DHA Selco (artemia enrichment), Sanocare Hatch Controller (artemia) |
| Vero Chemical/Pentair Aquatic Ecosystems | liquid chlorine, isopropyl alcohol,sodium thiosulfate/ Instant Ocean, Defoamer |
| Reed Mariculture | Algae Paste (rotifer feed/green water), Otohime dry feed (larval feed) |
| Western Chemical/Syndel | Ori-Green (rotifer enrichment)/ RAL Gun (Hormone injector), Oviplant (hormone) |
| SCI | Catheter tubing (sample fish eggs from females) |
| Biomark | Pit tags (tag broodfish) |
| Florida Aquafarms | Gelly Belly maturation diet premix |
End of presentation.
Go to top of page.