Live Feeds for Marine Fish Larvae

Live Feeds for Marine Fish Larvae
Aquaculture Center for Training, Education and Demonstration

Why live feeds?

This slide shows images of small fish larvae in water, demonstrating the tiny size and delicate nature of marine fish larvae that require live feeds for proper nutrition and development.
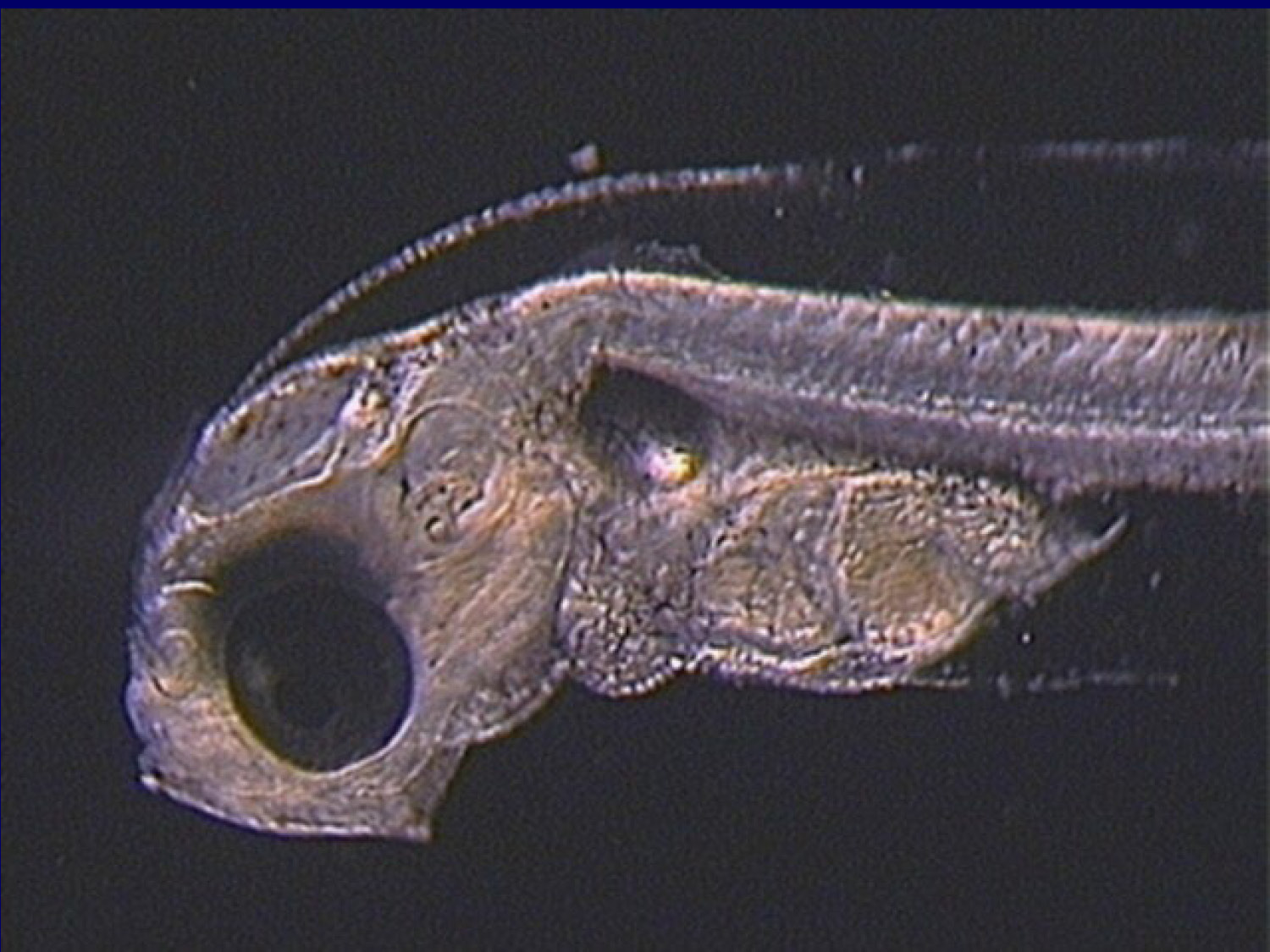
This slide shows an image of marine fish larvae, further illustrating the organisms that require live feeds.

Important Criteria For Selecting Live Feeds
- Is it the appropriate size for target species?
- Is the nutritional quality adequate for good growth and survival of the target species?
- Can it be produced cost effectively?
This slide shows three images of marine fish larvae.

Live Feeds
The two live feeds most commonly used are:
Rotifers (Brachionus sp.)
Artemia sp. (brine shrimp)
Other live feeds include: Molluscan trochophore larvae Ciliates
Copepods
Wild plankton

What makes good prey?
Size - prey size is dependent upon fish larvae mouth size. Typical initial prey size for most fish larvae is 50-120 microns in width.
Movement - slow moving prey are more easily captured by fish larvae.
Familiarity - fish larvae must learn to locate and capture prey. The more experience the larvae have with the prey, the higher the success rate.

This slide shows an image illustrating live feed organisms used in marine aquaculture.

Larval Feeding Strategies
Larval fish consume more food per unit body weight (50 – 300%) than fish in grow-out operations (2 – 10%)
Target Densities
1 to 10 prey per ml
Average Feeding Rates
200 to 600 prey per day
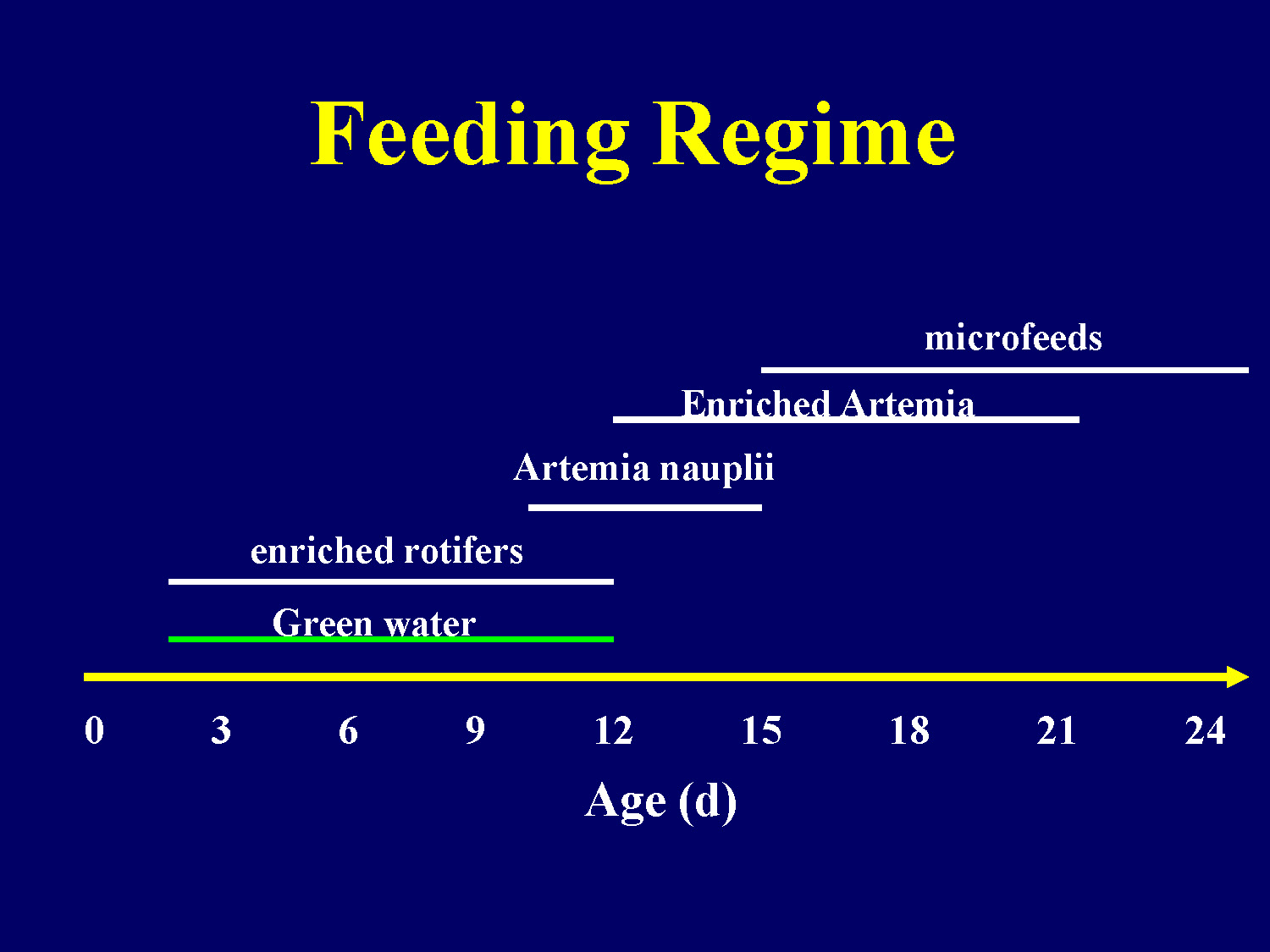
Feeding Regime
This chart shows the feeding schedule for fish larvae over a 24-day period (age in days). The feeding regime includes:
- Green water: from 0 to approximately 21 days
- Enriched rotifers: from 0 to approximately 15 days
- Artemia nauplii: from approximately 9 to 24 days
- Enriched Artemia: from approximately 15 to 24 days
- Microfeeds: from approximately 18 to 24 days
The x-axis shows age in days (0, 3, 6, 9, 12, 15, 18, 21, 24) and the different feed types are represented as horizontal bars across the timeline.
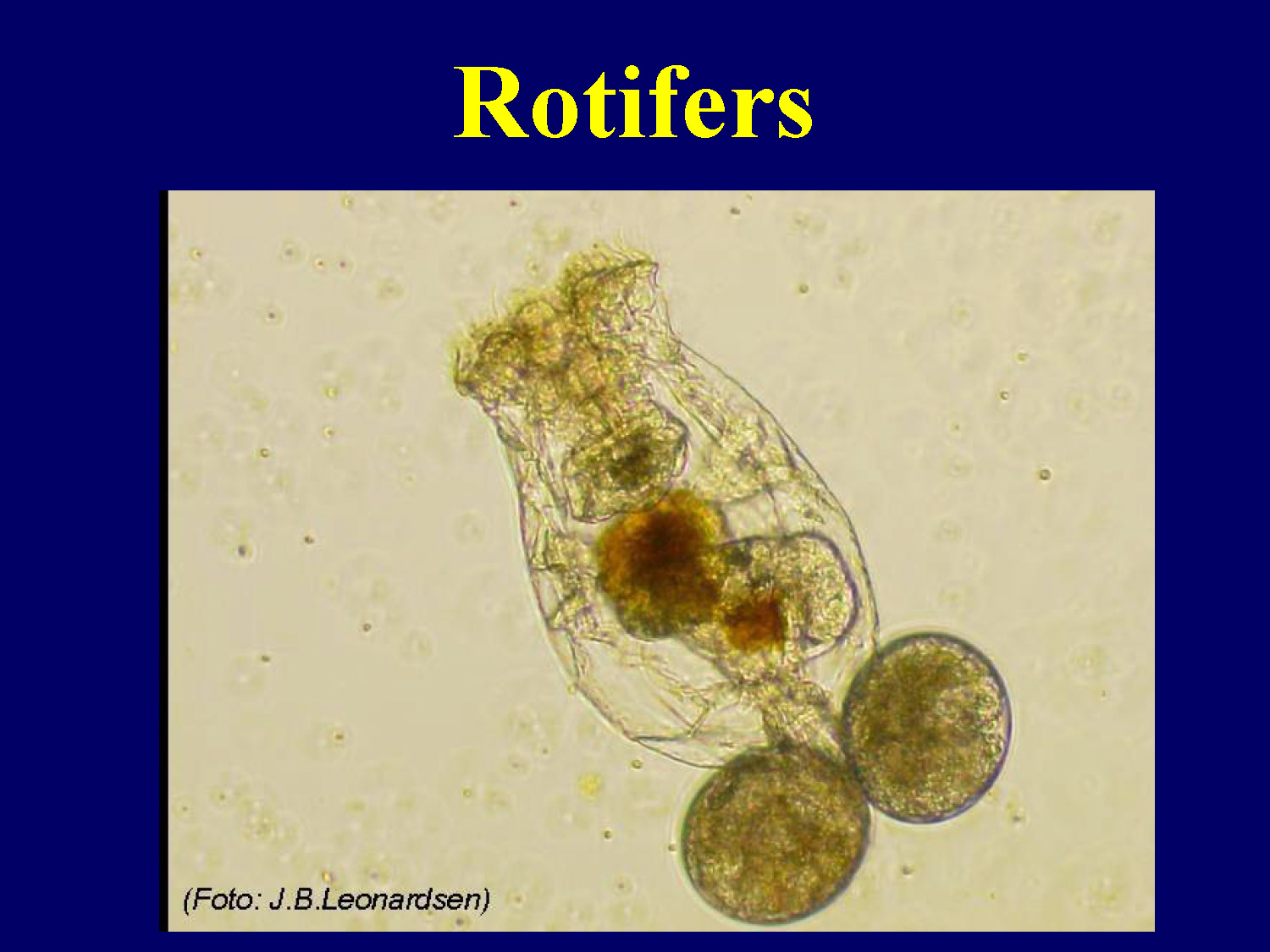
Rotifers
Image of Rotifer

Why use rotifers as live food?
Excellent for larval fish and shellfish
- High nutritional quality
- Small body size
- Slow swimming rate
- High rate of reproduction
- Low rate of mortality

What is a Rotifer?
- Microscopic protozoan
- Saltwater and fresh water environments
- Asexual and sexual reproduction
Phylum Rotifera "wheel-bearer" (> 2000 species)

Rotifer Corona
Cilia
This slide shows a microscope image of a rotifer with the corona (wheel-like structure) clearly visible, surrounded by cilia that create a rotating appearance when in motion.
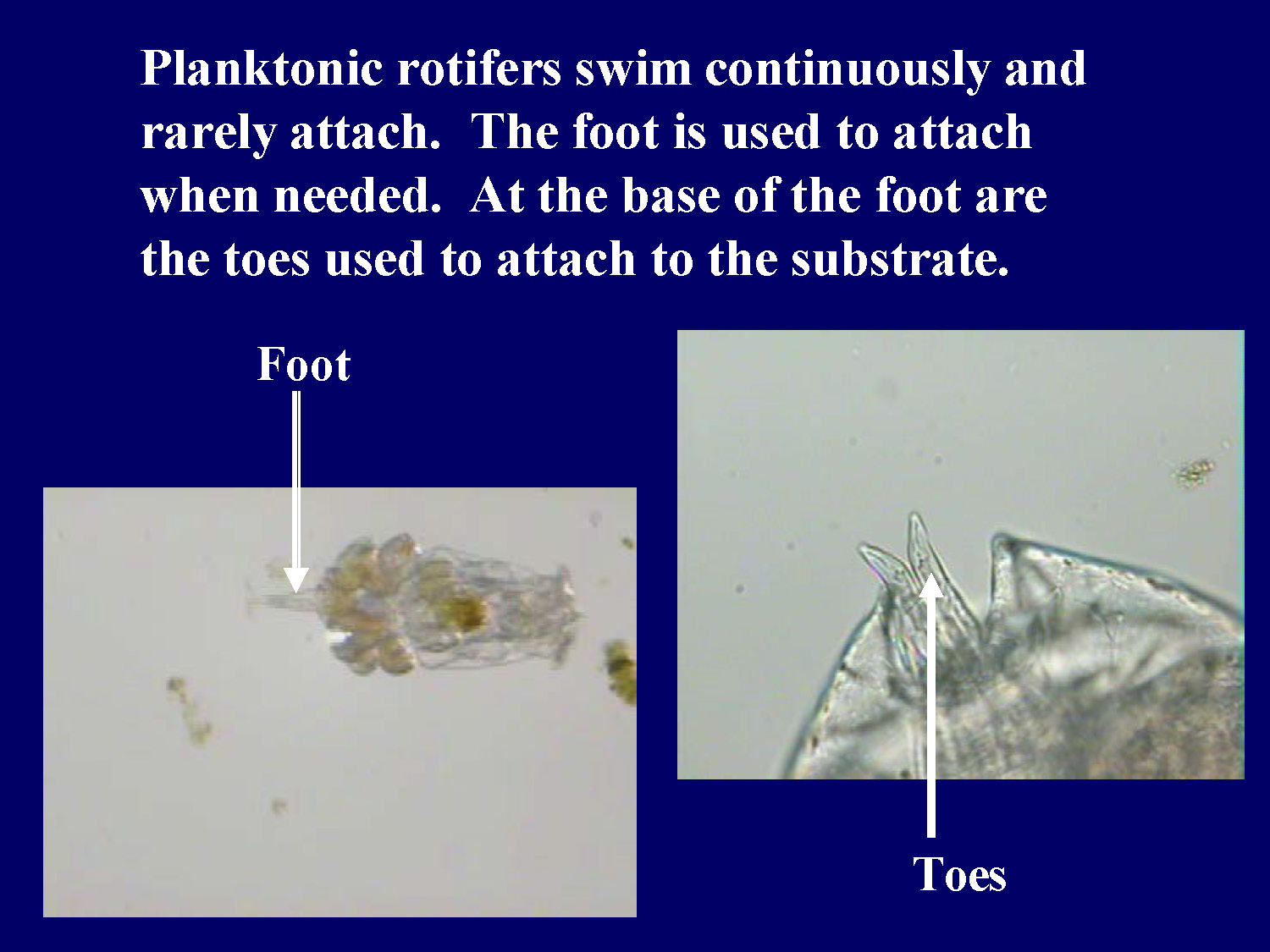
Planktonic rotifers swim continuously and rarely attach. The foot is used to attach when needed. At the base of the foot are the toes used to attach to the substrate.
Foot
Toes
This slide shows a microscope image of a rotifer with labels pointing to the foot and toes at the bottom of the organism.

Species & Strains
Marine Species
Brachionus plicatilis Brachionus rotundiformis
Freshwater
Brachionus rubens Brachionus calycflorus
Three Strains
L-type (large type, B. plicatilis)
Body length = 200 - 360 µm
S-type (small type , B. rotundiformis)
Body length = 150 - 220 µm
SS-type (special small, B. rotundiformis)
Body length = 70 - 160 µm

Optimal Conditions
Salinity below 35 ppt
Temperature range 27- 32°C
15 hour photoperiod
Moderate to low aeration
pH range from 6.5 - 8.0
Unionized ammonia concentrations: not to exceed 1 mg/liter
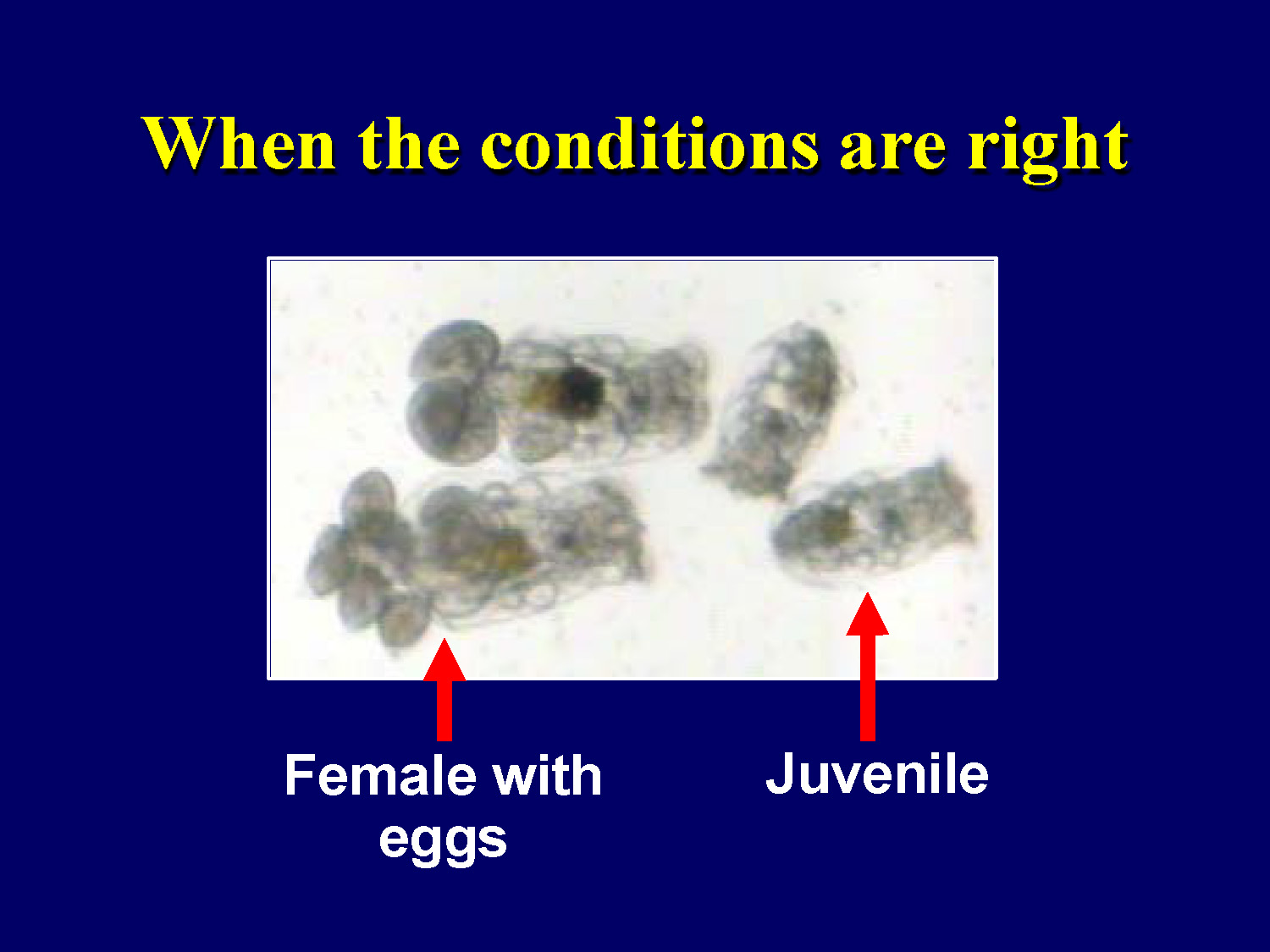
When the conditions are right
Female with eggs
Juvenile
This slide shows a microscope image of a female rotifer with visible eggs inside her body, demonstrating successful reproduction when environmental conditions are optimal.

Production Methods
- Cultures can be started by inoculations with live rotifers or by using cysts (resting eggs)
- Cysts have a shelf life of over one year
- Rotifers can be produced using algae, yeast, specialized products, or a combination
- Acceptable rotifer production ranges from 100 - 500 per ml
- Super-intensive culture requires specialized labor, equipment and techniques

Culture Methods
Continuous
Lasts 20 to 30 days
Harvest 20 to 30% per day
Refill with sterilized water daily
Batch (Our Method)
Harvest all rotifers at 3 to 5 days
Discard water
Re-stock with new water and rotifers

Intensive Production Methods
Requires:
- Continuous supply of algae
- De-foaming agents
- Filtration of water
- pH adjustments
- Oxygen gas
- Nylon filtration mats
Diligent & Constant Management!

Inside Rotifer Tank
This slide shows a photograph of the interior of a rotifer culture tank, displaying the water surface and culture equipment used in rotifer production.

This slide shows a photograph of rotifer culture equipment and facilities used in the production process.

Culture Condition
Rotifer cultures are healthy if:
- Rotifers are swimming rapidly
- Females with eggs : female ratio of about 1.2 :1
- Little or no bacteria or ciliates present
Cloudy water is a good sign of imminent crash
Crashing cultures will have a bad odor

This slide shows a photograph of rotifer culture tanks and equipment used in the production facility.

Our Rotifer Formula
Fill tank with seawater adjusted to 25 - 35 ppt
Add liquid chlorine (10%) & mix
Add sodium thiosulfate to neutralize chlorine and areate.
Add inoculating rotifers - a minimum of 150 to 250 per ml

Production Methods
Use light aeration
Keep light intensity to a minimum
Feed live algae/algae paste and rotifer diet daily
After 3 - 5 days rotifers will reach 500 to 750 per ml
Harvest and enrich for feeding and use to inoculate new tanks

Feeding Algae
Nannochloropsis oculata & Isochrysis galbana
Instant Algae
Live Algae
This slide includes images of commercial algae products used for feeding rotifers.

Rotifer Diets
This slide shows a photograph of various commercial rotifer diet products used in aquaculture facilities.

Rotifer Enrichment
Bioencapsulation – feed rotifers nutrients essential for larval fish
Enrichment increases the Omega 3 fatty acids in the rotifers.
Common enrichment products:
- Algae - live, frozen, or dried
- Emulsion products - liquid or dry
- Selco (culture and DHA protein), Microfeast, Algamac, RotiRich

Enrichment
What??
Why??
Fatty Acids
Amino acids
Vitamins
Pigments
Probiotics

Enrichments
20 million rotifers (10 am)
Water bath (28 °C) for 3 h
Harvesting
This flow chart shows the first steps of the enrichment process with arrows connecting each step.

Cold storage (5-10 °C)
This slide shows the final step of the enrichment process flow chart.

Rotifer Schedule
| 8:30 AM | Feed larvae enriched rotifers |
| 9:00 AM | Assess rotifer populations |
| 9:30 AM | Harvest tank, prepare enrichment containers, restart clean tank |
| 10:00 AM | Enrich rotifers(2-4 hour contact time) |
| 10:30 AM | Refill algae containers, AM rotifer feeding |
| 1:00 PM | Harvest, rinse, feed, and cold store rotifers |
| 5:00 PM | Feed larvae enriched rotifers |
| 9:00 PM | Feed larvae enriched rotifers |

Artemia
Photograph of Aremia salina

What is an Artemia?
Microscopic crustacean (brine shrimp)
Saltwater environments
Sold as cysts

This slide shows images of Artemia organisms used as live feeds in aquaculture.

Decapsulation
This diagram shows the decapsulation process:
Dehydrated cysts
Hydrated cysts
+ chemicals
The slide includes microscope images showing the different stages of the cysts.

Decapsulated cysts
This slide shows a microscope image of decapsulated Artemia cysts, which appear orange/light colored without their outer shell after chemical treatment.

Incubation
Sterilized water
25-30 g/L salinity
28-30 C
Vigorous aeration

15-20 h = hatching
"Umbrellas"
First instar
This slide shows microscope images of Artemia during hatching. The "umbrellas" are the empty cyst shells that float to the surface after hatching. The first instar nauplii are the newly hatched Artemia that appear 15-20 hours after incubation begins.

This slide shows a tank and equipment related to Artemia culture and production.

This slide shows a tank related to Artemia culture and production.

This slide shows images related to Artemia culture and production.

Enrichment
Second instar
This slide shows a microscope image of second instar Artemia nauplii, which are larger and more developed than first instar nauplii.

This slide shows images related to the Artemia enrichment process.

Sterilized water
25-30 g/L salinity
26-27 C
Moderate aeration
Enrichment contact 24 hrs.

This slide shows an image related to the Artemia enrichment process.

Artemia Schedule
| 8:30 AM | Feed larvae nauplii and/or enriched Artemia |
| 9-11:00 AM | Harvest Artemia tanks |
| 11:30 AM | Cold store harvested nauplii, start enrichment tank |
| 1:00 PM | Start next day enrichment, feed larvae |
| 5:00 PM | Feed larvae |
| 9:00 PM | Feed larvae |
Feedings should be spaced out during 24 period ~ 4-6 hrs.
This is the final slide of the presentation showing microscopic image of Artemia.
End of presentation.
Go to top of page.