Responsible Conduct of Research
Responsible conduct of research (RCR) is essential to good science. RCR promotes the aims of scientific inquiry, fosters a research environment that enables scientists to work together toward common goals, and promotes public confidence in scientific knowledge and progress for the public good.
However, there are several serious detrimental effects of research that is not conducted ethically and responsibly. These include, for example, data fabrication and falsification of results which undermine the pursuit of valid knowledge by misleading researchers to accept and further study false hypotheses or unreliable data. Likewise, plagiarism and harassment negatively impact the research environment by harming respect and trust among researchers. Fraudulent or socially irresponsible research undercuts the public’s trust and support for science.
Beyond preventing misconduct, RCR also promotes positive research behaviors, such as data transparency, proper authorship attribution, and the ethical treatment of human and animal subjects. Studies have shown that researchers who receive RCR training are more likely to adhere to these practices, contributing to higher standards of research integrity across institutions.
Training and education in RCR should occur not only during undergraduate or graduate studies, but throughout one’s research career. RCR education can take place in seminars, workshops, or conferences on research ethics, as well as through informal conversations during mentoring, training, and laboratory meetings about ethical conduct and practices.
RCR IN-PERSON WORKSHOPS
The Spring 2026 RCR Workshop series will take place from February 11th - April 14th on the FAU Boca Campus and on the MPFI Jupiter Campus. Space is limited, click here to register.
Establishment of what constitutes RCR are critical to assurance of research integrity. The Department of Health and Human Services’ Office of Research Integrity (ORI) has identified nine core elements of RCR: 1) the protection of human participants; 2) the welfare of laboratory animals; 3) conflicts of interest; 4) data management practices; 5) mentor and trainee relationships; 6) authorship and publication; 7) peer review; 8) collaboration; and 9) research misconduct. In addition to these elements, Florida Atlantic is committed to fostering safe research environments that are free from discriminatory harassment and expects researchers to engage as a responsible member of society through the practice of ethical science, understanding how their work has environmental and societal impact.
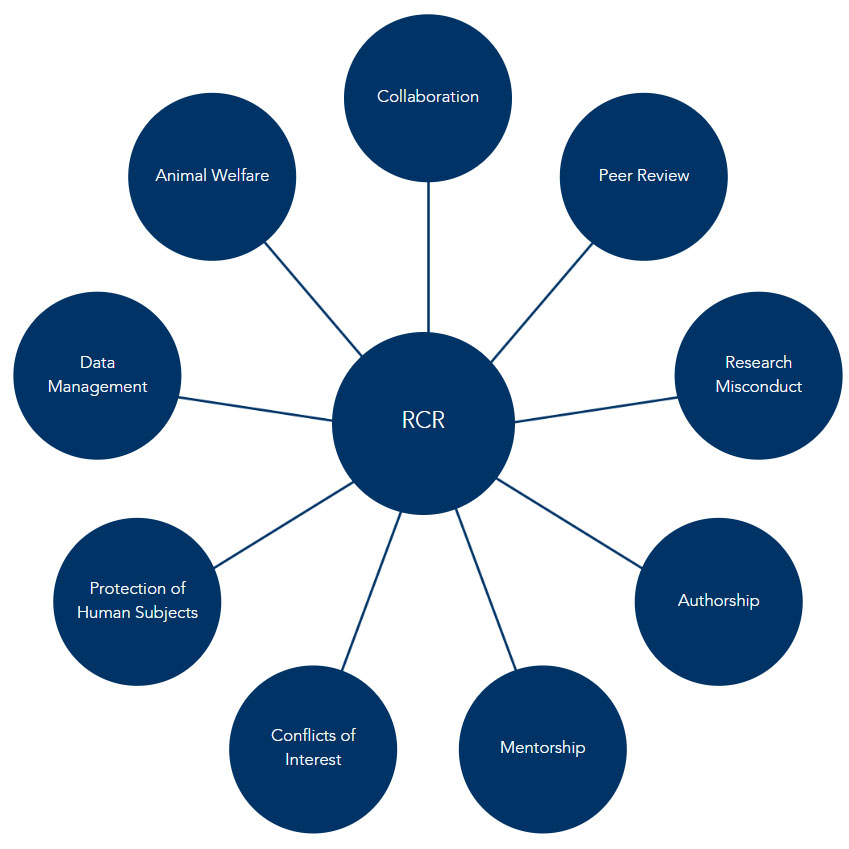
Collaboration:
This element of Responsible Conduct of Research concerns collaborations between different teams on a shared research plan and issues that may arise from such collaborations, both within and external to FAU. This includes setting ground rules, sharing materials, fair decision-making, and allocation of credit and authorship.
Peer Review:
This element of Responsible Conduct of Research concerns the purpose and importance of peer review in determining merit for research funding and publication. This includes participation in a Scientific Review Committee process (undergoing review or serving as a reviewer) and other formal and informal review activities, such as reviewing journal articles or colleagues' research proposals.
Research Misconduct:
This element of Responsible Conduct of Research concerns formal research misconduct as defined by regulations that govern research funded or conducted by the U.S. government. Formal research misconduct refers to plagiarism, fabrication, or falsification of research data; it does not refer to honest errors or differences of opinion.
Authorship:
This element of Responsible Conduct of Research concerns the purpose and importance of scientific publication and the responsibilities of authors. This includes assigning appropriate credit, acknowledgments, handling corrections and retractions, and managing the pressure to publish.
Mentorship:
This element of Responsible Conduct of Research concerns research mentors’ responsibilities to more junior researchers and to trainees in predoctoral and postdoctoral research programs. This includes the role of a research mentor, managing conflict, selection of a mentor, and appropriate boundaries of the mentor/mentee relationship.
Conflicts of Interest:
This element of Responsible Conduct of Research concerns the different types of conflict encountered by researchers and institutions and how to handle conflicts of interest. This includes identifying different sources of conflict (financial, professional, or personal) and appropriate strategies for disclosing, managing, and mitigating conflicts among various researchers and collaborators.
Protection of Human Subjects:
This element of Responsible Conduct of Research concerns the appropriate conduct of research involving human subjects. This includes differentiating between human subjects research and other activities (such as public health or program evaluation), ethical principles for human subjects research, and regulatory oversight.
Data Management:
This element of Responsible Conduct of Research concerns best practices for acquiring, analyzing, and maintaining research data. This includes appropriate methods of data collection, statistical analysis, data sharing, and storage in scientific research.
Animal Welfare:
This element of Responsible Conduct of Research concerns the appropriate conduct of research involving animals. This includes ethical principles for conducting research on animals, government regulations for animal research, institutional animal care and use committees (IACUCs), and humane treatment of animals.
This element of Responsible Conduct of Research concerns formal research misconduct as defined by regulations that govern research funded or conducted by the U.S. government. Formal research misconduct refers to plagiarism, fabrication, or falsification of research data; it does not refer to honest errors or differences of opinion.
This element of Responsible Conduct of Research concerns the purpose and importance of scientific publication and the responsibilities of authors. This includes assigning appropriate credit, acknowledgments, handling corrections and retractions, and managing the pressure to publish.
This element of Responsible Conduct of Research concerns research mentors’ responsibilities to more junior researchers and to trainees in predoctoral and postdoctoral research programs. This includes the role of a research mentor, managing conflict, selection of a mentor, and appropriate boundaries of the mentor/mentee relationship.
This element of Responsible Conduct of Research concerns the different types of conflict encountered by researchers and institutions and how to handle conflicts of interest. This includes identifying different sources of conflict (financial, professional, or personal) and appropriate strategies for disclosing, managing, and mitigating conflicts among various researchers and collaborators.
This element of Responsible Conduct of Research concerns the appropriate conduct of research involving human subjects. This includes differentiating between human subjects research and other activities (such as public health or program evaluation), ethical principles for human subjects research, and regulatory oversight.
This element of Responsible Conduct of Research concerns best practices for acquiring, analyzing, and maintaining research data. This includes appropriate methods of data collection, statistical analysis, data sharing, and storage in scientific research.
This element of Responsible Conduct of Research concerns the appropriate conduct of research involving animals. This includes ethical principles for conducting research on animals, government regulations for animal research, institutional animal care and use committees (IACUCs), and humane treatment of animals.
This element of Responsible Conduct of Research concerns collaborations between different teams on a shared research plan and issues that may arise from such collaborations, both within and external to FAU. This includes setting ground rules, sharing materials, fair decision-making, and allocation of credit and authorship.
This element of Responsible Conduct of Research concerns the purpose and importance of peer review in determining merit for research funding and publication. This includes participation in a Scientific Review Committee process (undergoing review or serving as a reviewer) and other formal and informal review activities, such as reviewing journal articles or colleagues' research proposals.
The importance of RCR education comes at a time when technology, bioengineering, bioinformatics, and translational research are changing research environs. Developments in precision medicine, organoid models, artificial intelligence, and availability of large datasets occur at basic science and clinical research points, making RCR translational and dynamic. These advances in research and discovery result in changing ethical landscapes and the need for continued education in conducting research responsibly.
The Florida Atlantic Division of Research (DoR) leads RCR programmatic efforts through both online and in person instruction, and coordinates with university personnel to ensure those participating in federally funded research or research training have the requisite RCR training.
To reinforce the concepts introduced in the online curriculum and in-person training, mentors and trainee(s) will be expected to meet regularly to discuss ongoing research, and the related ethical and procedural standards (i.e., data management, collaboration, authorship, etc.). Faculty and key personnel will be required to complete and maintain active RCR training as required by funding agencies and applicable institutional policies.
| Funding Mechanism | Required Training |
|---|---|
| NIH * | - Online RCR via CITI. - Eight (8) contact hours of live RCR education as stipulated by award. See below for details. |
| NSF | Online RCR via CITI and as stipulated in award or described in SOW. |
| Other Federal (Including passthrough) | - Faculty/ Key Personnel: As stipulated by sponsor or described in SOW - Students/ Post Doctoral Fellows: Online RCR via CITI |
| Other Sponsored Research | As stipulated by sponsor or described in SOW |
* NIH requires that all trainees, fellows, participants, and scholars receiving support through any NIH training, career development award (individual or institutional), research education grant, and dissertation research grant must receive instruction in responsible conduct of research. Applies to the following programs: D43, D71, F05, F30, F31, F32, F33, F34, F37, F38, K01, K02, K05, K07, K08, K12, K18, K22, K23, K24, K25, K26, K30, K99/R00, KL1, KL2, R25, R36, T15, T32, T34, T35, T36, T37, T90/R90, TL1, TU2, and U2R.
- https://grants.nih.gov/grants/guide/notice-files/NOT-OD-22-055.html
- https://www.niaid.nih.gov/research/responsible-conduct-research-training
- 10.1.1 Research Misconduct
- 10.1.2 Financial Conflict of Interest
- 10.1.6 Research Data
- Florida Atlantic Responsible Conduct of Research Program
Contact martinezj2012@fau.edu for additional questions regarding RCR training requirements