FAU Receives $1.7 Million NIH Grant for Novel Neuroinflammation Study
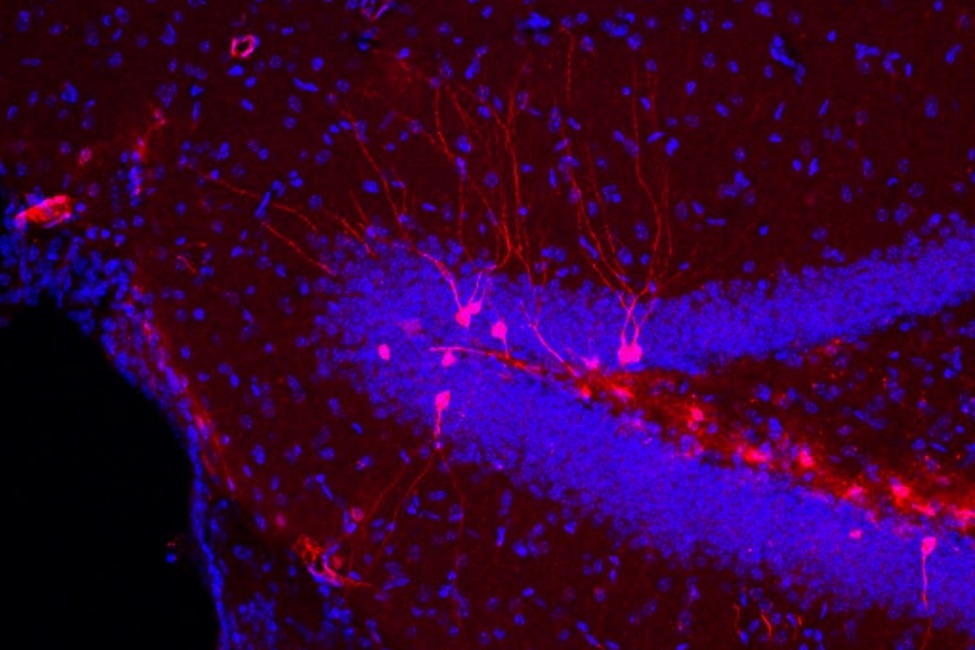
High power image of neurons (red) in the hippocampus of the mouse brain that express the IL-1 receptor (IL-1R1). Red color was generated by insertion of a fluorescent gene into the IL-1R1 gene. Both the round neuronal cell bodies and the thin, elongated dendrites that are characteristic of mature neurons can be seen. Blue color documents the presence of non-IL1R1 expressing neurons near ones that are IL-1R1 positive.
Anxiety, depression and cognitive decline are prominent psychopathologies that together affect more than 40 million people in the United States. These symptoms may manifest as co-morbidities of acute brain injury, chronic neurodegenerative diseases, central nervous system (CNS) autoimmune diseases, or as consequence of chronic stress, CNS infection, aging, or drug treatments with inflammatory molecules. The common thread among all these conditions is the presence of neuroinflammation.
Although neuroinflammation has recently been implicated as an important cause of these psychopathologies and a new target for the treatment of these conditions, the mechanisms underlying neuroinflammation-induced dysfunction of relevant neurocircuits remain poorly understood.
Neuroscientists from Florida Atlantic University’s Schmidt College of Medicine and Brain Institute (I-BRAIN) have received a five-year, $1.7 million R01 grant from the National Institutes of Health (NIH) for a project titled, “Neuroinflammation, Neuronal IL-1R1, and Behavior.” This novel project is the first to investigate how the inflammatory cytokine interleukin-1 (IL-1) influences neurotransmission through a direct action on neurons and how this action triggers behavioral changes.
“Many studies have suggested that elevated expression of IL-1 is one of the most critical links between neuroinflammation and psychopathology. Increased IL-1 expression, especially in the brain, has been observed in human patients with anxiety, depression, autism and/or cognitive deficits,” said Ning Quan, Ph.D., principal investigator, a professor of biomedical science in FAU’s Schmidt College of Medicine, and a member of FAU’s I-BRAIN. “Evidence strongly suggests that IL-1 could be a mediator of anxiety, depression and learning/ memory deficits. However, we lack a good understanding of the precise neuronal circuits through which IL-1 affects neurons and how the receptor signals to induce these psychopathologies.”
Quan and co-investigator Randy Blakely, Ph.D., executive director of FAU’s I-BRAIN and a professor of biomedical science, FAU's Schmidt College of Medicine, will establish nIL-1R1 as a crucial link that could convert neuroinflammation to neural dysfunction, providing a new pathogenic mechanism for anxiety, depression, and cognitive dysfunction. For the study, they will focus on the specific sets of neurons that express IL-1R1, identifying them as the vulnerable targets to this pathogenic mechanism. Results from their research could suggest new targets for the treatment of psychopathology.
“Very few studies have attempted to investigate how IL-1 directly modulates neuronal activity and little is known about the function of neuronal IL-1 receptor in the central nervous system,” said Quan. “This missing gap can be attributed to the inability to identify IL-1mediated responses. In addition, because nIL-1R1 is expressed at low levels, detection by traditional immunohistochemical methods is difficult.”
To overcome these technical limitations, Quan and Blakely recently generated a mouse system to both visualize and express IL-1R1 in specific cell types in the brain or to eliminate its expression in these cells to understand how IL-1R1 loss influences behavior.
For the project, the researchers will pursue three specific aims: map and characterize IL-1R1 expressing neurons in the brain to provide the first brain map of nIL-1R1 distribution; determine the role of neuronal IL-1R1 in the induction of behavioral deficits and neuropathology caused by neuroinflammation; and elucidate the cellular and molecular pathways by which neuronal IL-1R1 causes psycho- and neuro-pathology. The research team recently reported in the journal Immunity that IL-1R1 is expressed at high levels by a limited set of neurons in the brain and have recently observed that sub-inflammatory levels of brain IL-1 appear to selectively activate nIL-1R1 on these cells whereas higher levels of IL-1 are required to activate non-neuronal cells.
“Florida Atlantic University neuroscientists are on the cutting-edge of developing genetic tools and techniques to help researchers pinpoint mechanisms that contribute to some of the most debilitating neurodegenerative diseases that affect us globally,” said Blakely. “With this latest grant from the National Institutes of Health, we hope to elucidate the key signaling pathways of a key neuroinflammatory pathway linked to behavioral deficits and neuropathology following neuroinflammation – a pathological feature of a wide range of central nervous system diseases.”


-FAU-
Tags: faculty and staff | science | medicine | research | jupiter