Malaria Runs Like Clockwork; So Does the Parasite Causing the Disease
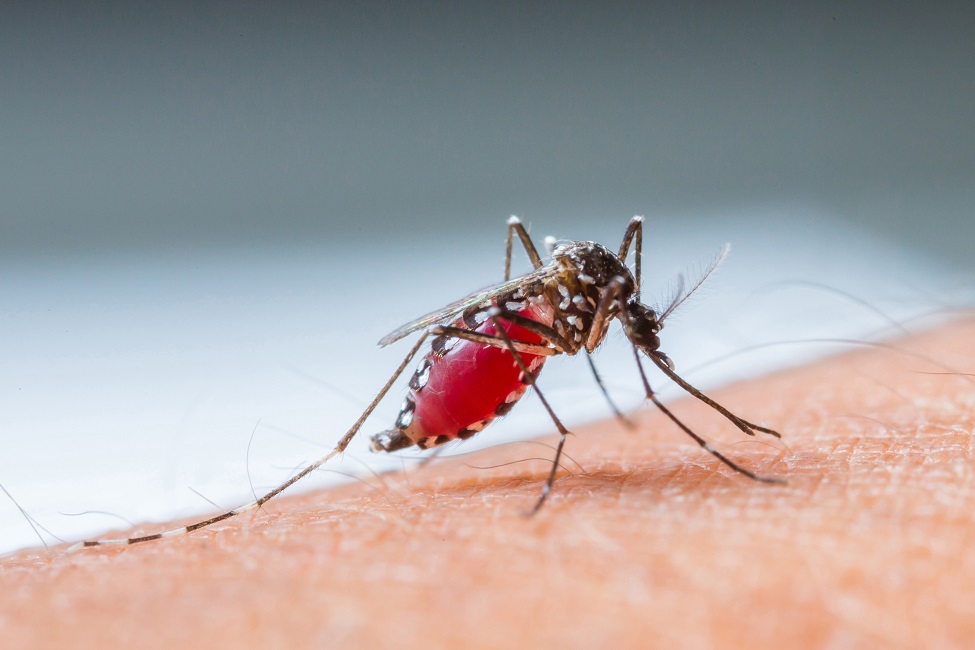
The malaria parasite is transmitted by female Anopheles mosquitoes, which bite mainly between dusk and dawn.
In humans, malaria is caused by the genus Plasmodium. The malaria parasite is transmitted by female Anopheles mosquitoes, which bite mainly between dusk and dawn. During one stage of infection in the human host, successive broods grow inside red blood cells and destroy them, releasing daughter parasites – “merozoites” – that continue the cycle by invading other red cells. The blood stage parasites are those that cause the symptoms of malaria.
Malaria runs like clockwork. Patients infected with the deadliest species, P. falciparum, often exhibit fever cycles, and these cycles coincide with the blood stage of the infection where the parasite progresses. The blood-stage infection of the malaria parasiteexhibits a 48-hour developmental cycle that culminates in the synchronous release of parasites from red blood cells, triggering 48-hour fever cycles in the host. The human-infecting species of Plasmodium repeat this cycle every 24, 48, or 72 hours (depending on the species), suggesting that cycles could be driven by a host circadian cycle or a parasite-intrinsic oscillator with circadian periodicity.
Scientists have long speculated the possibility that the parasites themselves might have their own internal clock. While there are many examples of the importance of circadian or daily rhythms in host-parasite interactions, thesourceof the Plasmodium’s rhythmic life cycle is a central, unsolved question.
Francis Motta, Ph.D., second author and an assistant professor in the Department of Mathematical Sciences at Florida Atlantic University’s Charles E. Schmidt College of Science, is among a team of scientists who have uncovered evidence that an intrinsic oscillator drives the blood stage cycle of the malaria parasite, P. falciparum. Results of the study, just published in Science, suggest parasites have evolved mechanisms to precisely maintain periodicity.
For the study, Motta and collaborators from Duke University, Walter Reed Army Institute of Research, and Montana State University investigated the rhythmic behavior of the P. falciparum intraerythrocytic cycle in an in vitro culture system where canonical circadian signals from the host were not present. Using high-density time-series transcriptomics and microscopy for four strains of P. falciparum, they compared several key molecular features of these cycles with molecular signatures produced by circadian networks and eukaryotic cell-cycle oscillators.
“Malaria and its causal parasite, the Plasmodium genus, are fundamentally rhythmic entities. Our study has shown that the parasite has molecular signatures common to known circadian and cell-cycle oscillators,” said Motta. “Each of the four strains we examined has a unique period, indicating strain-intrinsic period control. We also demonstrate that parasites have low cell-to-cell variance in a cycle period, on par with a circadian oscillator.”
-FAU-
Tags: research | faculty and staff | science